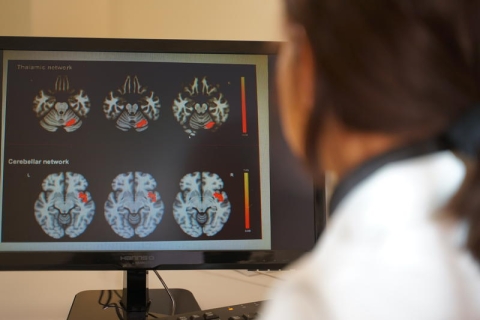
Multiple sclerosis (MS) is a chronic, inflammatory, autoimmune disease of the central nervous system (CNS) characterized by progressive deterioration of myelin, the “envelope” ensheating neuronal processes. Myelin damage leads, in turn, to neuronal dysfunction, impairment of nerve-to-nerve communication and progressive neurological disabilities. While myelin damage is rather efficaciously repaired during the first stages of the disease - thanks to the presence, in the adult CNS, of immature oligodendrocyte precursor cells (OPCs) – during MS chronicisation myelin repairing ability (“remyelination”) is progressively lost, and patients experience more and more severe and irreversible neurological symptoms. Currently available treatments mainly control inflammation and modulate patients’ immune response, but no remyelinating drugs are available yet.
The paper by Parravicini and coll., that appeared in these days in PLoS ONE, reports the in vivo myelin protecting activity of galinex, a synthetic compound that specifically acts on GPR17, a receptor expressed by OPCs.
«Several years ago, we discovered that activation of GPR17 on cultured OPCs by agonist ligands induced cells to start maturation and produce myelin, raising the hypothesis that selective GPR17 compounds could favor remyelination in MS, thus reinstalling nerve impulse transmission» says pharmacologist Prof. Maria Abbracchio at University of Milan where research on GPR17 was started. «But» she adds, «to prove that this could effectively occur in vivo, we strongly needed to develop a highly specific agonist for the GPR17 receptor».
«To this aim» says Prof. Ivano Eberini, a computational biochemist at the same University «we developed an iterative drug discovery pipeline, where more than 1,000,000 chemical compounds were virtually tested in silico on a structural model of GPR17. This approach started from the identification of 3 diverse families of ligands that, in collaboration with colleagues at University of Pisa and Aptuit in Verona, were then “exploded" - i.e., varied by in silico combinatorial chemistry and refined, for subsequent testing on recombinant cells expressing GPR17. The best scoring molecules on the receptor were then subjected to testing on OPC cultures, followed by analysis on OPC/neuronal cocultures to evaluate their ability to promote myelin formation, and, finally, selected for in vivo testing in experimental autoimmune encephalomyelitis, an established rodent model of MS».
The rodent experiments were designed by Chiara Parravicini, first author on the paper, together with the molecular biologists and in vivo pharmacologists Davide Lecca and Davide Marangon. “One of the best GPR17 agonist, galinex, had shown promising pharmacokinetic and metabolic properties in in silico simulations, and was for this reason chosen for testing in rodents” says Chiara Parravicini. «As predicted» she declares, «galinex proved able to significantly retard the appearance of symptoms during disease induction in mice».
This study was funded by the Italian Multiple Sclerosis Foundation which has been supporting research of Professor Maria Pia Abbracchio’ on the role of the GPR17 receptor in neurodegenerative syndromes for 10 years.
«The multi-year collaboration between the Italian MS Foundation and the University of Milan has led to this important result,» says Paola Zaratin, director of scientific research of the Foundation, «which opens the way for new molecules to treat progressive forms of disease».
Referenza
Titolo: Development of the first in vivo GPR17 ligand through an iterative drug discovery pipeline: A novel disease-modifying strategy for multiple sclerosis.
Autori: Parravicini C, Lecca D, Marangon D, Coppolino GT, Daniele S, Bonfanti E, et al. (2020)
Rivista: PLoS ONE 15(4): e0231483.
Doi: https://doi. org/10.1371/journal.pone.0231483